 This process (which is called a chain reaction) escalates very rapidly. 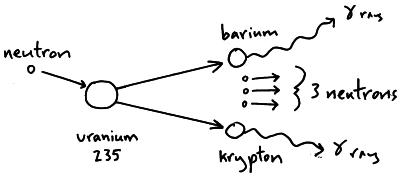 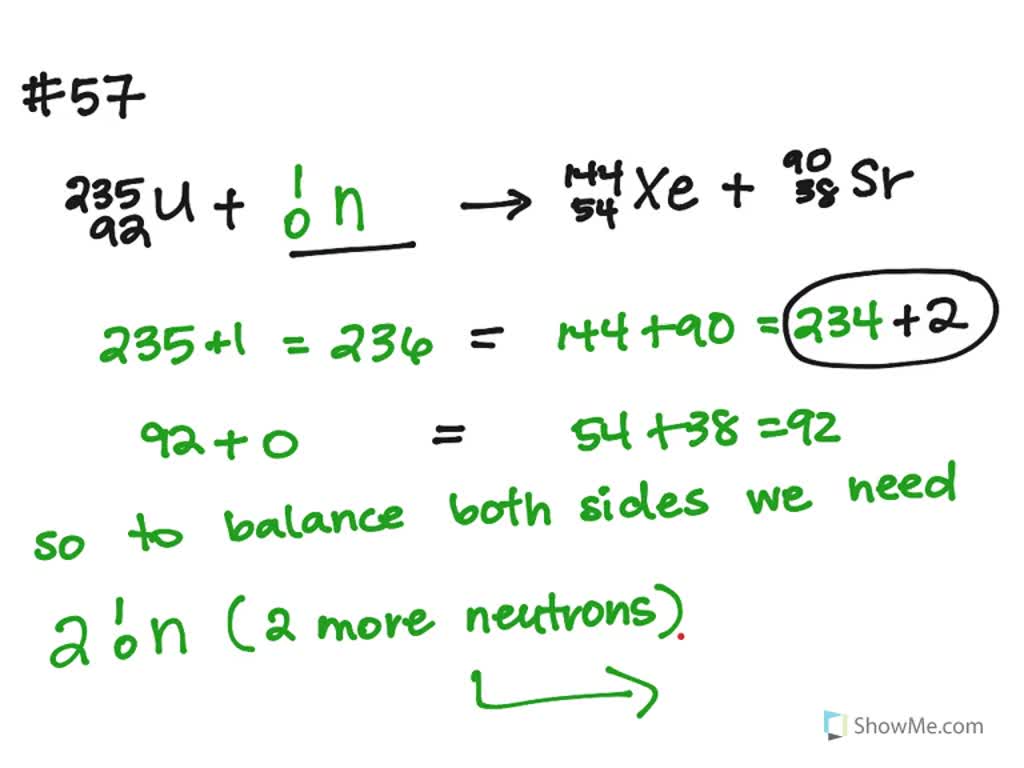
A third repetition will produce 27 neutrons. These in turn will fission three more U-235 nuclei, producing a total of nine neutrons. For instance, when one mole of U-235 undergoes fission, the products weigh about 0.2 grams less than the reactants this lost mass is converted into a very large amount of energy, about 1.8 × 10 10 kJ per mole of U-235. As soon as a stray neutron bombards a U-235 nucleus, fission will take place and three neutrons will be produced. You notice that $57+42 \neq 92$, if that was the case, it would be equal, but I don't clearly see where the difference physically comes from and what to add or subtract (and why) from to the first or from the second term to get the other result. Plutonium-239 (239 Pu or Pu-239) is an isotope of plutonium.Plutonium-239 is the primary fissile isotope used for the production of nuclear weapons, although uranium-235 is also used for that purpose. A tremendous amount of energy is produced by the fission of heavy elements. this made possible a chain reaction with an unprecedented energy yield. One of these neutrons is needed to sustain the. 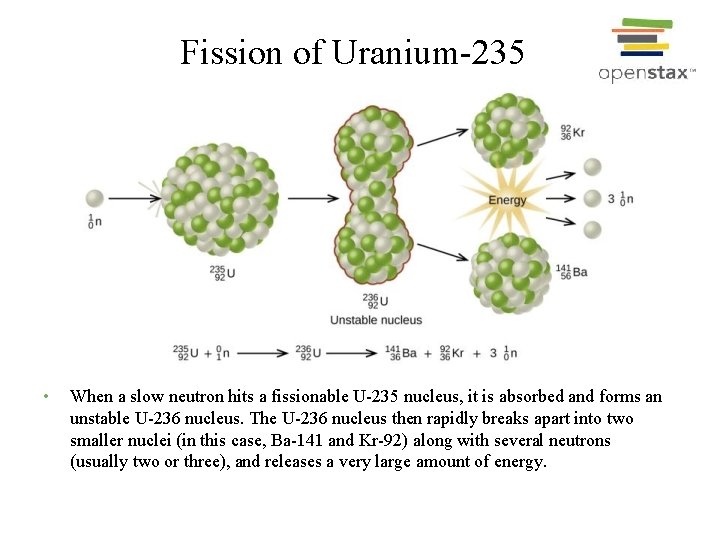
The Science Of Nuclear Energy Week 1 3 What Is Fission Openlearn Open University. that the U-235 fission yielded an enormous amount of energy. Fission of U-235 nuclei typically releases 2 or 3 neutrons, with an average of almost 2.5. What Are All The Possible Products Of A Reaction Where Uranium 235 Undergoes Nuclear Fission Socratic. Consider the neutron induced fission $\text Nuclear Fission Equation 2 Examples Practical Guide Linquip. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
